Suppressing Ras Gain of Function Mutations
|
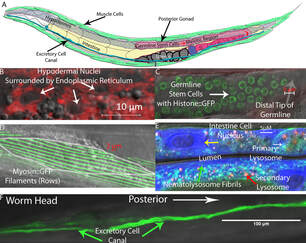
Our current project is focused on determining how animals bearing the mutant Ras oncogene are able to live without developing neoplasias. Our approach to understanding this has three aims. First, we identify genes that increase or decrease the penetrance and expressivity of a particular Ras allele. Concurrently, we directly observe cell divisions happening in animals with the Ras gain of function mutation to determine what happens differently in the animals that do not develop neoplasias. Finally, we alter the dosages of genes that affect the penetrance of the Ras allele to determine exactly how cell cycle and cell fate transformation processes are affected.
|
Intrinsic Noise Control (Stochastic Allele Expression Bias Control)
|
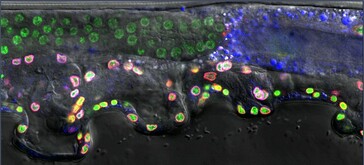
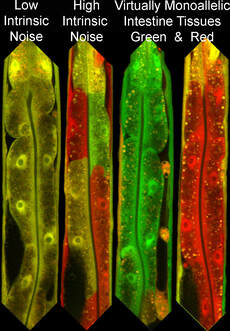
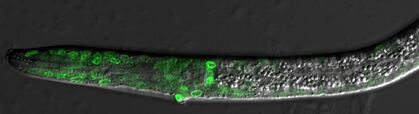
Our project on intrinsic noise control is focused on understanding how biological systems control stochastic autosomal allele biases. We refer to autosomal allele biases as intrinsic noise. We determine the amount of intrinsic noise for each particular gene by measuring each cells' deviation from biallelic expression among cells sampled from a given tissue. Each gene has its own particular intrinsic noise set point under standard conditions. Genes with high intrinsic noise appear variegated, with some cells showing biallelic expression and others showing expression skewed towards a single allele. In some animals, we see entirely monoallelic cells within a tissue. However, genetic and environmental differences can cause the intrinsic noise level of a gene to change. We want to control these biases for specific genes to affect human health and disease outcomes. Our research aims to identify cis and trans control elements to define an intrinsic noise control pathway.
|
Environmental Toxicants and Intrinsic Noise
|
|
Our research on environmental toxicants and intrinsic noise aims to determine how environmental toxicants (human-made pollution) affect autosomal allele biases in metazoans. This is a completely unexplored area of research combining systems biology and toxicology to understand how environmental toxicants may be affecting the natural amounts of intrinsic noise in specific genes. Consequences for environmental toxicants on intrinsic noise may include allele-specific silencing or inappropriate expression of both alleles of a gene that is supposed to be biased or monoallelic. This research is also developing allele-specific sequencing technologies with the Seelig and Kennedy Labs and the UW EDGE Center.
|
Aging and Intrinsic Noise
Proximal Causes of Aging
|
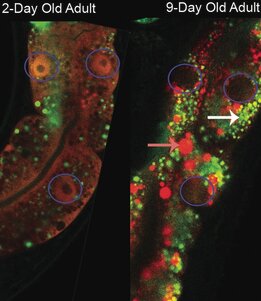
In this project, we wish to determine which organelles fail first in cells comprising the distinct tissues in aging C. elegans. Determining how metazoan cells fail with age and how these intracellular failures affect organ function and organismic physiology are critical to our understanding of the aging process. For example, we have determined that gene expression becomes dysregulated before lysosomes begin failing. This is different from what has been observed in brewer's yeast, the only organism for which we have a cause of age-related death at the cellular level.
|
Determining How Diapause Exit Mechanisms Rejuvenate Somatic Cells
|
|
When aged nematodes exit adult diapause, they rejuvenate their soma. They increase the volume of cells comprising their tissues and clear stalled lysosomes filled with fluorescent proteins. We do not know what other organelle changes happen during aging in diapaused adults, but we are excited to find out how these animals are able to effectively rejuvenate their cells.
|